Another great post from Salvador Herrando Pérez.
—
Commercial and sport fishing establish minimum body sizes for catches of many species to preserve fish stocks. Recent work reveals that sustainable fisheries also depend on the regulation of the harvest of the biggest fish, at least in long-lived species.
Growing up in Spain in the 1980s, I was taken by a Spanish television spot featuring a shoal of little fish sucking colourful dummies, and at the same time (how they managed, I never questioned) singing the motto Little fish? No, thanks. The then Ministry of Agriculture, Fishery and Food created this media campaign to create awareness among consumers not to buy immature fish at local markets – “…a 60-gram hake will only weigh 2 kg after two years” the add stated.
Indeed, the regulation of fish harvest by age classes is substantial to any fishery. In particular, the protection of younger fish has been a beacon of fishery policy and management that dates back to the 19th century when, among others, the British ichthyologist Ernst Holt concluded that: “…it is desirable that fish should have a chance of reproducing their species at least once before they are destroyed” 1. Very much in line with such principles, conventional fish stock management has in practice neglected the mature age classes2, other than for the fact that they are the end point of extraction and what we consumers eat on the table.
Fisheries management has traditionally relied on manipulating the take of juvenile and adult fractions, and how harvest-driven mortality, environmental variability and density-dependent processes (especially cannibalism, competition, disease and predation) affect the demography of those two broad age classes. A consequence of such a dichotomous vision is a dichotomous choice of managing one or the other; in fact, the juvenile fraction has been historically championed by far3.
In modelling fishery dynamics, many mathematical equations put emphasis on the same premise, e.g., the pioneering and still influential stock-recruitment model4 by Ricker, and the Beverton and Holt model5. Alas, Beverton and Holt5 contended that the most compensatory density feedback must occur at the juvenile stage in the majority of fish species, so management options should aim to maximise the survival of the younger classes. In the last decade, however, a different perception has grown since fishery scientists have started to dive deep into the interactions between fishing and life history of exploited species from several angles3,6-9. One of those angles highlights “the importance … of leaving the big ones” 10,11.

Adult and larvae of rock fish (Sebastes melanops). Following a short gestation, females release hundreds of thousands juveniles equipped with a fat globule (red arrow). Each juvenile must forage from external sources (initially plankton) before exhausting their fat reservoir. Rock fish inhabit the shelf bottoms from California to Alaska and the Aleutian Islands in the Western Pacific. Larvae are pelagic, and adults are benthic down to ~ 400 meters and prey on other fish (S. Lonhart/NOAA & C. Chapman).
An illustration of this concept is found in the paper led by the late Steven Berkeley on the reproduction of black rockfish (Sebastes melanops) off Oregon, USA12. This species is fairly long-lived (< 50 years), big (< 5 kg), and viviparous (gives birth to live young), so the eggs spawn within the mother’s body where they receive nourishment for about a month. Mothers then release up to one million larvae (< 1 cm), each venturing into the pelagic environment loaded with a little tummy-pack of fat. Mortality rates peak at this stage of the life cycle.
Berkeley found that larvae born from older females had larger fat reservoirs, grew quicker and withstood better the lack of food than those from younger mothers (Figure 1). At sea, those features should increase survival until the young learn to find food by themselves. In a complementary study, Bobko and Berkeley13 also revealed that age differences up to ten years can triple the number of fertilized embryos from younger to older rockfish females.
The gamble of reproduction
Fish species channel their reproductive effort through three main strategies14: opportunistic species have a short life, grow quickly and produce many offspring (e.g., anchovies); equilibrium species are long-lived, grow slowly and produce few young (e.g., sharks); and periodic species are also long-lived and grow slowly, but produce many young (e.g., rock fish) [notice that salmonid15 and intermediate16 strategies have also been described].
For equilibrium and periodic species, a long life often implies a large body. In terms of fitness, as an individual grows and ages, its chances of successful reproduction at least once in a life time increase, as though reproduction resembled a lottery whereby the older managed to bet more frequently than the younger (one side of the gamble). Equilibrium species bet a few descendants at each reproductive bout, whereas periodic species bet from thousands to millions. If reproduction fails one year, the long-lived individual can wait for improved conditions in the next year, e.g., more food, more mates, better environmental conditions.
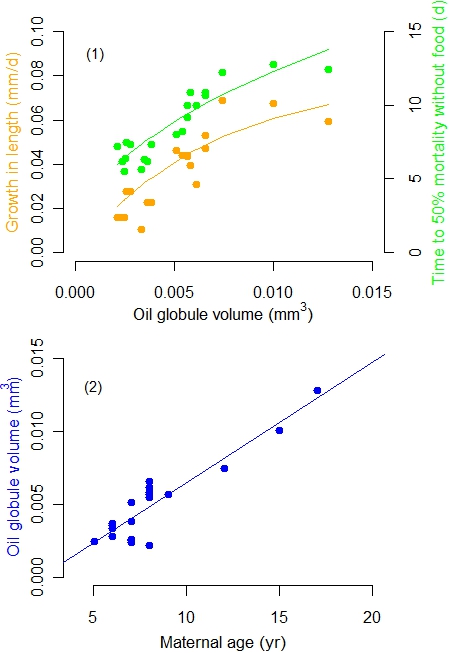
Fig. 1: Reproductive traits of rock fish (Sebastes melanops) examined in larvae from wild females caught off Oregon (USA).
Many multi-million dollar fisheries target relatively long-lived species, and it will be no surprise to find them on the menu or delicatessen section of our favourite restaurant or shop (e.g., tuna, cod, halibut, caviar [i.e., sturgeon]). That many harvested fish stocks worldwide show a temporal trend of decreasing body size17-19 indicates not only the selective removal of the largest fish, but also rapid evolutionary responses to compensate for (to escape from!) big-fish mortality, like retarding body growth and advancing the age at first reproduction20,21.
The handicap of fishing already age-truncated stocks is that their fluctuations are magnified relative to untouched populations18,19. So the other side of the gamble comes along: many poor-fishing years ensue, which makes everybody unhappy in the long-term, from the fishers through to the managers and consumers.
Anderson et al.19 have recently tested three hypotheses to explain why stock fluctuations are magnified by intensive fishing:
- population shift could track shifts in harvest pressure (unsupported)
- truncation of age structure by harvest leads to predominance of small/young fish unable to buffer environmental fluctuation because their fitness is lower than that of large, long-lived fish (weakly supported), and
- individuals’ vital rates in those populations with few large individuals are more variable (strongly supported).
Whatever the mechanism, if we target big fish then stocks become unpredictable and, what might be even more shocking, if we target only small or only big fish, the result might be population depletion all the same22,23. Thus, conservation of age structure seems to be the key issue. At an ecosystem level (beyond the coverage of this article), when overharvesting concentrates not only on the largest individuals within a species, but on the species that reach the largest sizes, i.e., apex predator species, we are actually ‘fishing down marine food webs’ 24 – one of the main anthropogenic impacts on global fisheries25.
The main take-home message from Berkeley’s and other studies is that the assessment of stock recovery and exploitation must rely not only on biomass, but also on maximum age. Fish populations and communities lacking their giants risk depletion and extinction.
References
- Holt, E. W. L. An examination of the present state of the Grimsby trawl fishery, with especial reference to the destruction of immature fish. Journal of the Marine Biological Association of the UK 3, 337-446 (1895).
- Longhurst, A. Murphy’s law revisited: longevity as a factor in recruitment to fish populations. Fisheries Research 56, 125-131, doi:10.1016/s0165-7836(01)00351-4 (2002).
- Lorenzen, K. Fish population regulation beyond “stock and recruitment”: the role of density-dependent growth in the recruited stock. Bull. Mar. Sci. 83, 181-196 (2008).
- Ricker, W. E. Stock and recruitment. Journal of the Fisheries Research Board of Canada 11, 559-923 (1954).
- Beverton, R. J. H. & Holt, S. J. On the dynamics of exploited fish populations. Vol. 19 (Ministry of Agriculture, Fisheries and Food , UK, 1957).
- Jennings, S., Reynolds, J. D. & Mills, S. C. Life history responses to fisheries exploitation. Proceedings of the Royal Society of London Series B-Biological Sciences 265, 333-339 (1998).
- Rose, K. A., Cowan, J. H., Winemiller, K. O., Myers, R. A. & Hilborn, R. Compensatory density dependence in fish populations: importance, controversy, understanding and prognosis. Fish and Fisheries 2, 293-327 (2001).
- Winemiller, K. O. Life history strategies, population regulation, and implications for fisheries management. Canadian Journal of Fisheries and Aquatic Sciences 62, 872-885 (2005).
- Denney, N. H., Jennings, S. & Reynolds, J. D. Life-history correlates of maximum population growth rates in marine fishes. Proceedings of the Royal Society of London B 269, 2229-2237, doi:10.1098/rspb.2002.2138 (2002).
- Birkeland, C. & Dayton, P. K. The importance in fishery management of leaving the big ones. Trends in Ecology & Evolution 20, 356-358, doi:10.1016/j.tree.2005.03.015 (2005).
- Berkeley, S. A., Hixon, M. A., Larson, R. J. & Love, M. S. Fisheries sustainability via protection of age structure and spatial distribution of fish populations. Fisheries 29, 23-32, doi:10.1577/1548-8446(2004)29[23:fsvpoa]2.0.co;2 (2004).
- Berkeley, S. A., Chapman, C. & Sogard, S. M. Maternal age as a determinant of larval growth and survival in a marine fish, Sebastes melanops. Ecology 85, 1258-1264, doi:10.1890/03-0706 (2004).
- Bobko, S. J. & Berkeley, S. A. Maturity, ovarian cycle, fecundity, and age-specific parturition of black rockfish (Sebastes melanops). Fishery Bulletin 102, 418-429 (2004).
- Winemiller, K. O. & Rose, K. A. Patterns of life-history diversification in North American fishes: implications for population regulation. Canadian Journal of Fisheries and Aquatic Sciences 49, 2196-2218 (1992).
- McCann, K. & Shuter, B. J. Bioenergetics of life history strategies and the comparative allometry of reproduction. Canadian Journal of Fisheries and Aquatic Sciences 54, 1289-1298 (1997).
- King, J. R. & McFarlane, G. A. Marine fish life history strategies: applications to fishery management. Fisheries Management and Ecology 10, 249-264, doi:10.1046/j.1365-2400.2003.00359.x (2003).
- Hutchings, J. A. & Reynolds, J. D. Marine fish population collapses: Consequences for recovery and extinction risk. Bioscience 54, 297-309, doi:10.1641/0006-3568(2004)054[0297:mfpccf]2.0.co;2 (2004).
- Hsieh, C. H. et al. Fishing elevates variability in the abundance of exploited species. Nature 443, 859-862, doi:10.1038/nature05232 (2006).
- Anderson, C. N. K. et al. Why fishing magnifies fluctuations in fish abundance. Nature 452, 835-839, doi:10.1038/nature06851 (2008).
- Conover, D. O. & Munch, S. B. Sustaining fisheries yields over evolutionary time scales. Science 297, 94-96, doi:10.1126/science.1074085 (2002).
- Olsen, E. M. et al. Maturation trends indicative of rapid evolution preceded the collapse of northern cod. Nature 428, 932-935, doi:10.1038/nature02430 (2004).
- Schröder, A., Persson, L. & de Roos, A. M. Culling experiments demonstrate size-class specific biomass increases with mortality. Proceedings of the National Academy of Sciences 106, 2671-2676, doi:10.1073/pnas.0808279106 (2009).
- Kooten, T. v., Persson, L. & Roos, A. M. d. Size-dependent mortality induces life-history changes mediated through population dynamical feedbacks. The American Naturalist 170, 258-270 (2007).
- Pauly, D., Christensen, V., Dalsgaard, J., Froese, R. & Torres, F. Fishing down marine food webs. Science 279, 860-863, doi:10.1126/science.279.5352.860 (1998).
- Baum, J. K. & Worm, B. Cascading top-down effects of changing oceanic predator abundances. J. Anim. Ecol. 78, 699-714, doi:10.1111/j.1365-2656.2009.01531.x (2009).




[…] Oceans need their giants (02/11/11,CB) / Overfishing large-bodied fish […]
LikeLike
[…] bird losses, taxonomic inflation, niche conservatism, historical biogeography, ecological traps and ocean giants) has produced a post this week expanding on the problem of roads. Also weirdly coincidental is that […]
LikeLike
[…] makes an appearance inRelated BlogsRelated Blogs on Divers take dead aim at invasive fish speciesOceans need their giants « ConservationBytes.com© 2011, . All rights reserved. Posts Related to Divers take dead aim at invasive fish species […]
LikeLike
Great article, just a shame it takes eons to move both fisheries managers and fishers to thinking differently. I see fisheries scientists pushing the age structure concept in fisheries, but it’s so hard because they’re always seen as the bad guys. At least I know now and can push this from another angle. Thanks, Salvador!
LikeLike