
Brent geese flock in the Limfjorden (Denmark) – courtesy of Kevin Clausen. The Brent goose (Branta bernicla) is a migratory goose that breeds in Arctic coasts, as well as in northern Eurasia and the Americas, starting from late May to early June. Adults are about 0.5 m long, weigh some 2 kg and live up to 30 years. Their nests are placed in the ground, where reproductive pairs incubate a single clutch (≤ 5 eggs) for a couple of months. They are herbivores, feeding on algae (mainly Zostera marina in Limfjord) and seagrass in estuaries, fjords, intertidal areas and rocky beaches during fall and winter. During summer they feed on tundra herbs, moss, lichens, as well as aquatic plants in rivers and lakes. The species is ‘Least Concern’ for the IUCN, with a global population at some 600,000 individuals.
Migratory birds synchronise their travel from non-breeding to breeding quarters with the seasonal conditions optimal for reproduction. Above all, they decide when to migrate on the basis of the climate of their wintering areas while they are there. As climate change involves earlier springs in the Arctic but not in the wintering areas, there is a lack of synchronisation that leads to a demographic decline of these birds in the polar regions where they breed.
When I think about how species respond to climate change, the song from the Clash “Should I stay or should I go” comes to mind. As climate changes, species eventually have to face an ultimate choice: (i) stay and adapt to novel conditions or become locally extinct if adaptation fails, (ii) or move to other regions where climatic conditions should be more suitable. Migratory species have to face this decision every time they have to move back and forth from non-breeding to breeding grounds.
Migration is a behavioural strategy shared by different animal groups like sea turtles, mammals, amphibians, insects or birds. Species move from one area to another usually to feed and reproduce in the best climatic conditions possible. For birds, migration is a common phenomenon that typically entails large movements between breeding and wintering grounds. These vertebrates boast some of the longest migratory distances known in the animal kingdom, particularly seabirds like Artic terns, which can complete up to a round-world trip in a single migratory event between the UK and the Antarctic (1). There are several theories about the mechanisms triggering bird migration, including improving body condition and fitness through unexploited resources (2), reducing parasite load (3), minimizing predation risk (4), maximizing day-light (5), or reducing competition (6, 7). Whatever the cause, birds have to decide when the best moment to migrate is, counting only with the (usually climatic) clues they have at the departure site.
Anyone who uses public transportation will be familiar with missing a train or a bus that has departed earlier than usual. The consequence: late arrival at destination. Nowadays our smartphones alert us of changes in transportation schedules and we can easily adjust to those changes. In nature, many species are able to synchronize the best time to migrate or hibernate in response to climatic conditions. The synchrony is subject to natural selection generation after generation, and determines the reproductive success of many migratory bird species, because their arrival to the breeding grounds should match in time and space with food availability and a climate benign for reproduction (8). But this biological clock is breaking for many species because of climate change (9).
An extreme example takes place in the Arctic. The Arctic climate offers a narrow window of opportunity to raise offspring for many migratory birds — so timing is critical for success. To make it harsher for Arctic wildlife, the Arctic spring has been advancing in the last few decades relative to North and Central Europe (10), thus challenging the breeding success of many migratory bird species. Danish scientists Kevin and Preben Clausen have studied the effect of this seasonal asynchrony in pale-bellied Brent goose (Branta bernicla hrota) between 1989 and 2012 (11). A population of these geese spends the winter in the North Sea, mainly in Denmark, and every May migrates to their polar breeding grounds in the Svälbard Archipelago (Norway), after a 3000 km, non-stop flight. During their trip they navigate over the Lista Bird Observatory (south of Norway) where around 60% of the geese population can be tracked annually.
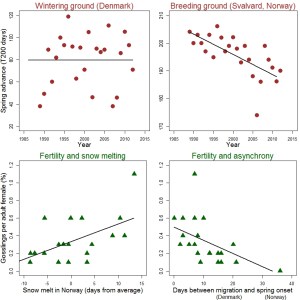
Climate and reproduction of brent geese migrating from Limfjorden (Denmark), where they accumulate fat during winter, to the Svalbard archipelago (Norway), where ~ 7600 individuals breed in spring and summer (11). Red circles show the onset of the spring over three decades, which has occurred gradually earlier only in the breeding grounds. Spring onset is measured in T200 days = annual cumulative number of days up to 200 degrees Celsius (ºC) resulting from adding temperature averages only in days > 0 ºC from 1 Jan. Lower T200 represents earlier spring. Green triangles show geese fertility against snow melting and seasonal asynchrony. Fertility correlates positively with melting time and negatively with asynchrony magnitude. Melting dates estimated as the number of days for the snow layer to disappear in Svalbard (lower the value, earlier the melting), and asynchrony estimated as the difference between the onset of migration in Denmark and T200 in Svalbard (larger the difference, larger the asynchrony).
Clausen and Clausen found an approximately constant spring onset in Denmark during the 24 years of study, with the geese starting to migrate within an interval of 2 to 7 days in late May. However, spring has advanced more than two weeks in Svälbard during the same stretch of time, so the geese have been arriving at the archipelago gradually earlier relative to the onset of spring. As a consequence, they are failing to exploit the Arctic food chains, which activate immediately after the winter. Additionally, during the study period, the Clausens found that the proportion of juveniles in the population had decreased from 15% in the 1980s to 10% in the 2000s. Such a drop resulted from increased juvenile mortality in response to earlier ice melting in Svälbard, and the growing asynchrony between departure times in Denmark and the start of the Arctic spring. As the annual natural mortality of this population is 13%, its long-term viability is no longer guaranteed.
Long migrants lack environmental clues to gauge both the occurrence and magnitude of seasonal asynchronies between temperate and polar latitudes driven by modern climate change. If such asynchrony was spatially homogeneous (i.e., earlier spring both in wintering and breeding grounds), its impact on the fitness of migratory species could be lower. The phenomenon is affecting many other migratory birds (9,12) unable to predict lags in peaks of food (13,14) and predators (15), or habitat loss (16). We should keep an eye on these species to prevent their population collapse, while they stand as ideal model organisms to understand how anthropogenic climate change is to shape the distribution of zoological biodiversity on Earth.
—
by Salvador Herrando-Pérez @hp_salva & David R. Vieites @vieites, Museo Nacional de Ciencias Naturales
(with support of the British Ecological Society and the Spanish Ministry of Economy, Industry and Competitiveness)
References
- Egevang C, et al. (2010) Tracking of Arctic terns Sterna paradisaea reveals longest animal migration. Proceedings of the National Academy of Sciences of the USA 107: 2078-2081
- Berthold P (1988) Evolutionary aspects of migratory behavior in European warblers. Journal of Evolutionary Biology 1: 195–209
- Piersma T (1997) Do global patterns of habitat use and migration strategies co-evolve with relative investments in immunocompetence due to spatial variation in parasite pressure? Oikos 80: 623-631
- McKinnon L, et al. (2010) Lower predation risk for migratory birds at high latitudes. Science 327: 326–327
- Schekkerman H, et al. (2003) Mechanisms promoting higher growth rate in arctic than in temperate shorebirds. Oecologia 134: 332–342
- Cox GW (1968) The role of competition in the evolution of migration. Evolution 22: 180-192
- Safriel UN (1995) The evolution of Palearctic migration — the case for southern ancestry. Israel Journal of Zoology 41: 417-431
- Carey C (2009) The impacts of climate change on the annual cycles of birds. Philosophical Transactions of the Royal Society B: 364: 3321-3330
- Miller-Rushing AJ et al. (2010) The effects of phenological mismatches on demography. Philosophical Transactions of the Royal Society B 365: 3.177-3.186
- Hoye TT et al. (2007) Rapid advancement of spring in the High Arctic. Current Biology 17: 449-451
- Clausen KK, Clausen P (2013) Earlier Arctic springs cause phenological mismatch in long-distance migrants. Oecologia 173: 1101-1112
- Saino N et al. (2011) Climate warming, ecological mismatch at arrival and population decline in migratory birds. Proceedings of the Royal Society B 278: 835-842
- Van der Jeugd HP et al. (2009) Keeping up with early springs: rapid range expansion in an avian herbivore incurs a mismatch between reproductive timing and food supply. Global Change Biology 15: 1057-1071
- Wood EM, Kellermann JL (2015) Phenological Synchrony and Bird Migration: Changing Climate and Seasonal Resources in North America. CRC Press, Boca Raton (Florida, USA)
- Madsen J, et al. (1998) Correlates of predator abundance with snow and ice conditions and their role in determining timing of nesting and breeding success in Svalbard light-bellied brent geese Branta bernicla hrota. Norsk Polarinstitutt Skrifter 200: 221-234
- Clausen KK, et al. (2012) Energetic consequences of a major change in habitat use: endangered Brent geese Branta bernicla hrota losing their main food resource. Ibis 154: 803-814



[…] from our children to grandparents, posing fast adaptive challenges to all species (see here, here and […]
LikeLike
[…] Spring asynchrony in migratory birds (15/05/17,CC) / Seasonal mismatch […]
LikeLike