
The term ‘ecology’ in 16 different languages overlaid on the oil on board ‘The Tower of Babel’ by Flemish Renaissance painter Pieter Bruegel the Elder (1563).
In his song ‘Balada de Babel’, the Spanish artist Luis Eduardo Aute sings several lyrics in unison with the same melody. The effect is a wonderful mess. This is what the scientific literature sounds like when authors generate synonymies (equivalent meaning) and polysemies (multiple meanings), or coin terms to show a point of view. In our recent paper published in Oecologia, we illustrate this problem with regard to ‘density dependence’: a key ecological concept. While the biblical reference is somewhat galling to our atheist dispositions, the analogy is certainly appropriate.
—
A giant shoal of herring zigzagging in response to a predator; a swarm of social bees tending the multitudinous offspring of their queen; a dense pine forest depriving its own seedlings from light; an over-harvested population of lobsters where individuals can hardly find reproductive mates; pioneering strands of a seaweed colonising a foreign sea after a transoceanic trip attached to the hulk of boat; respiratory parasites spreading in a herd of caribou; or malaria protozoans making their way between mosquitoes and humans – these are all examples of population processes that operate under a density check. The number of individuals within those groups of organisms determines their chances for reproduction, survival or dispersal, which we (ecologists) measure as ‘demographic rates’ (e.g., number of births per mother, number of deaths between consecutive years, or number of immigrants per hectare).
In ecology, the causal relationship between the size of a population and a demographic rate is known as ‘density dependence’ (DD hereafter). This relationship captures the pace at which a demographic rate changes as population size varies in time and/or space. We use DD measurements to infer the operation of social and trophic interactions (cannibalism, competition, cooperation, disease, herbivory, mutualism, parasitism, parasitoidism, predation, reproductive behaviour and the like) between individuals within a population1,2, because the intensity of these interactions varies with population size. Thus, as a population of caribou expands, respiratory parasites will have an easier job to disperse from one animal to another. As the booming parasites breed, increased infestations will kill the weakest caribou or reduce the fertility of females investing too much energy to counteract the infection (yes, immunity is energetically costly, which is why you get sick when you are run down). In turn, as the caribou population decreases, so does the population of parasites3. In cybernetics, such a toing-and-froing is known as ‘feedback’ (a system that controls itself, like a thermostat controls the temperature of a room) – a ‘density feedback’ (Figure 1) is the kind we are highlighting here.
Figure 1 – Three examples of density feedback
 |
(a) Spanish imperial eagle (Aquila adalberti) from Mediterranean scrubland (Doñana National Park) recovered from long-term poaching in the 1950s. As the population expanded by the mid-1970s, competition for decreasing high-quality nesting sites reduced fertility (compensatory density feedback). The population experienced stability at ~15-16 pairs in the 1970s and 1980s, but adult mortality increased in the 1990s. As a result, increased matings involving less-fertile juvenile mates decreased reproductive rates (depensatory feedback)27. Photo V. Penteriani. |
 |
(b) Land transformation (agriculture, roads) confined Good’s banksia (Banksia goodii) to 16 known populations in Albany by the 1990s. Smaller patches had lower fertility (depensatory feedback) likely because of inbreeding failure and reduced visits by native pollinators (honey possums, honeyeaters)28. Photo B. Lamont. |
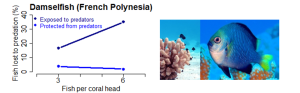 |
(c) Yellow-tailed damselfish (Dascyllus flavicaudus) use corals for shelter in Moorea (French Polynesia). Holbrook and Schmitt29 transplanted coral heads at two fish densities with and without protection (nets) from predatory fish. Mortality increased competition for coral shelter from low to high fish density (compensatory density feedback). Photo M. Holbrook-Schmitt. |
Because the magnitude with which individuals are eaten by predators or compete for food, space or other resource (along with other factors such as climate, genetics or life history) determines how fast a population grows or declines, density-dependent relationships have wide applications in ecosystem management and conservation. Most of these are apparent at the highest and lowest ends of the spectrum of population abundances. At high numbers, if a fraction of a population dies off naturally due to limited food resources, killing the surplus of individuals should theoretically have a minor impact on a target population’s growth rate, and estimates of that surplus (= quotas) can guide strategies of predator protection, hunting4 and fishery limits5. At low numbers, measuring minimum populations sizes at which deaths outnumber births can be critical to establish a species’ conservation status, protected areas, harvest moratoria, predator releases for pest control or invasion thresholds2,6. This link between density feedback and population persistence and extinction has made the concept prominent in important fields such as conservation biology7, biological control8 or fishery management5.
Terminological caveats
To ground their views in their publications, scientists use words and mathematical expressions; but count the number of words and equations in any publication, words always win! Unfortunately, when we use words in a style subject to no real rules (apart from grammatical, although science writing is increasingly fraught with terrible grammar), the odds for distorting the ecological vocabulary are fairly high. Indeed, the outcome of the last 100 years of demographic research is an impressive collection of more than 60 different terms to name DD relationships!
From the 1950s to recent times, experts have strived to tidy up this ever-growing jargon9,10. The epic of the task was immortalised by George Varley10 who stated in 1958 that “biologists do not apply the term ‘chromosome’ to any coloured body”. The situation has provoked numerous confrontations in the specialised literature (much of which is indigestible for the average ecologist), and has consequently impacted communication among ecologists within and across taxa and disciplines. Herein we summarise the causes and consequences of terminological inconsistency of DD – which we fully elaborate in our article in Oecologia.
We detected 4 scenarios causing proliferation of jargon, as follows.
‘Capsicum’ and ‘bell pepper’ are the same thing (synonymy)
The effect of increasing population size on a demographic rate can either enhance or arrest population growth. Therefore, we essentially need two terms (not 60!) to define both types of relationship. Unfortunately, different authors have given different names to those two relationships, because they believed their terms were better than existing ones, or because they were unaware that the concept’s key terms had already been coined elsewhere. This situation partly reflects the problem that many scientists work secluded in their specialised areas of expertise.
The first DD terms for cases where population size arrests population growth were by the American entomologist Harry Smith11, who referred to ‘density-dependent mortality factors’ where predators kill more individuals of an insect pest as the pest expands; and by the Japanese botanists Kichiro Shinozaki and Tatuo Kira12, who coined ‘self-thinning’ where, as trees planted in an area of land grow in biomass, individuals end up competing for resources (light, nutrients) and some gradually die as a result. Thereafter, other authors created conceptually similar terms like: ‘centripetality’ (in the field of wildlife management), ‘compensation’ (botany, entomology, fisheries), ‘direct DD’ (protozoology), ‘disoperation’ (social biology), ‘intensity dependence’ (parasitology), ‘negative DD’ (genetics) or ‘negative feedback’ (systems theory).
The American social biologist Warder Allee13 branded the first DD terms for cases where population size boosts population growth. He referred to ‘inverse density-dependent factors’ where cultivated protozoans benefitted from cooperative interactions as the group grew in members; and by the Canadian fisheries biologist Ferris Neave14, who defined ‘depensatory mortality factors’ (Neave actually invented the term ‘depensatory’) where, for juvenile salmon migrating from their native river to the sea, the more compact the school of juveniles, the fewer the losses to predators. Likewise, other authors created synonyms like ‘cooperation’ (social biology), ‘positive DD’ (genetics), ‘underpopulation’ (entomology) or ‘positive feedback’ (systems theory).
‘Pear’ also means ‘banana’ (polysemy)
Researchers have re-invented the definition of existing DD terms, indicating that they did not read or take into consideration the foundational literature where the terms were originally coined and defined. This problem fits a general trend of neglect of older literature in which many hypotheses and theories of our modern science were laid out many decades ago15. With regard to DD, three terms have become intensively polysemous, namely ‘negative’, ’positive’ and ‘direct’.
A population can decline if an increase in population size causes an increase in mortality and emigration (positive relationship) and/or a decrease in fertility, survival and immigration (negative relationship). What is important here is that statistical relationships of different sign (positive or negative) contribute to the same effect on the population change (decline). The British geneticist Jack Haldane16 referred to ‘positive’ and ‘negative’ DD as any relationship between population size and a demographic rate that contributes to population boom or decline, respectively. The terminological confusion has emerged when ecologists have named the sign of the statistical relationships, but not the density effect on population change. For instance, if an increase in the number of limpets on a rock over summer causes increased mortality due to competition for space (positive correlation), many would call that ‘positive’ DD, but the effect is population decline, i.e., ‘negative’ DD according to Haldane’s terminology.
A different situation has occurred with the term ‘direct’ (in maths, ‘direct’ is, by definition, a positive relationship between any two variables) as opposed to ‘inverse’ (a negative relationship) DD. Allee coined those terms to differentiate negative and positive correlations between population size and mortality. Thereafter, the two terms have suffered two severe modifications. First, ecologists expanded their use to name effects rather than statistical relationships (i.e., ‘direct’ for DD [on any type of demographic rate] whereby higher densities leads to reduced population growth rate, and ‘inverse’ for cases where density increases lead to increasing population growth rates). Second, disregarding Allee’s foundational use, many authors now mean ‘immediate’ to refer to ‘direct’ DD for density feedback measured between two consecutive sampling times (e.g., eagles, cats, snakes and many other predators feed intensely on a population boom of a grassland rodent, so ironically only a few rodents are left to reproduce next year), as opposed to ‘delayed’ DD over more than two sampling times (e.g., the booming rodent lives on an island with one fox species [which has longer generation time than the rodent] as its only predator, so it might take several years until the predator population increases enough to reduce rodent numbers).
Modifying the letters in ‘apple’ to spell ‘leppa’ or ‘ppale’ does not equate to the original meaning (inflation)
Most ecologists would agree with DD being defined as “…a dependence of per capita population growth rate on present and/or past population densities”17. However, behind such a simple definition lies an enormous body of controversial research that (in simple terms) has contrasted the demographic roles of biotic interactions (as inferred from DD) versus abiotic factors like climate. The topic would deserve an entire dissertation (which we summarise in our Oecologia paper to stress that ‘density dependence’ does not mean ‘population regulation’, and that much semantic and terminological inflation has originated from authors emphasising population change in time versus space). The point at issue is that, to explain their personal views, debating authors have created a battery of new terms with a strong philosophical flavour, such as ‘ineffective’, ‘explicit’, ‘phenomenological’, ‘weak’, ‘Nicholsonian’ or ‘classical’ DD. We believe that these controversies, and the resulting jargon, have impeded, rather than progressed, ecology. In particular, if we accept those terms created to distinguish DD that can be detected statistically and DD that truly underpins a demographic process, then we should be ready to do the same with any other statistical relationships with potential biological significance, e.g., is there statistical and causal pollination, or global warming, etc.? The distinction relates to statistical reporting and inferences rather than to terms and concepts.
‘Golden’ and ‘Red Delicious’ differentiate types of ‘apple’ (refinement)
As we progress in our understanding of how nature works, new concepts and new terms to name them are genuinely needed. For instance, the concept/term ‘Allee effect’ represents a particular case of ‘inverse’ DD13, only at low population numbers. In the last two decades, ecologists have refined our understanding of the measurement and meaning of Allee effects, hence producing new qualifiers like ‘dormant’, ‘multiple’, ‘non-additive’ or ‘strong’ – indeed, 17 terms are defined by Berec et al.18. ‘Allee effects’ stand for mechanisms only activated as populations decline to near extinction (e.g., by over-harvest or the starting point of a recently established invader). Those mechanisms are, for instance, that individuals cannot find conspecifics (too few to find over a large area) of the same sex (too many males for too few females, or vice versa), the dismantling of cooperative interactions (e.g., hunting or offspring rearing becoming inefficient as group size shrinks) or inbreeding depression (reduced fitness through genetic similarity). The magnitude of the former mechanisms is magnified by further population declines into a vortex of extinction. Obviously, this concept has enormous importance in biodiversity conservation and management2,6.
Some consequences
It goes without saying that the messy triplet (synonymy, polysemy, terminological inflation) is a dominant feature of the ecological literature. This triplet becomes apparent when experts publish reviews of the history, evolution and involution of particular terms, e.g., ‘habitat’, ‘niche’ or ‘carrying capacity’, or one is at a conference where two speakers (apparently) disagree or talk about different topics because they are using a different lexicon.
Terminological inconsistency is indeed a serious problem at different levels:
- Terminology through intellectual dispute reflects, not concepts, but schools of thoughts; yet terminology driven by opinion rather than empirical/theoretical facts contributes little to scientific progress, as illustrated by the long-standing debate around population regulation19
- Poor terminological standards trouble students and early-career researchers trying to understand and apply concepts on which the very experts disagree20 – the legacy of individual authors also relies on how understandable their published work is to the younger (but future) generation of scientists. I (Corey) remember as a lowly MSc student that I couldn’t quite get my head around the difference between ‘regulation’ and ‘limitation’, for exactly these reasons! I (Salva) spent hours of at the beginning of my PhD in search of a (non-existent) density-independent regulation in opposition to the common expression “density-dependent regulation”.
- A regular activity of any researcher is to browse databases for papers (SCI, ASFAS, Scopus), and we all be familiar with searches by key-word that yield hundreds to thousands of papers, many of which are not relevant to our target query
- More generally, English is taken as the rule (certainly the international one) for scientific communication, but many researchers do not comprehend, or have access to appropriate training in English – it is not surprising that publication rates across countries can be strongly driven by language21 (well, if you are English-speaking, try to write a paper in Italian or French, even if you are lucky to have funds for a translation). Certainly, the understanding and expression of scientific information by non-English speakers is seriously jeopardized by inconsistent terms – even the translation of specialised treatises or textbooks can be problematic because translation relies on context rather than terms – and… how do we translate a term created in English into Spanish or Arabic, or vice versa?
- The implementation of environmental policies across transnational jurisdictions can be challenged when country-based terminological frameworks conflict; for instance, Dauvin et al.22 satirised that we need ‘mental gymnastics’ to classify marine habitats according to the European Habitat’s Directive, in which terms and definitions reflect the rival English and French schools of thought, e.g., about what a habitat or ecotone means
- Finally, ecologists are increasingly interacting with society through policy makers, management and conservation planners and the media23; therein, scientific discourse becomes a tool of communication with non-scientists, and clear terminology is instrumental to important matters like funding applications, and conveyance of scientific information for societal benefit
Controlled terminology is a reflection of scientific maturity
Ecologists use terminology the way they do because the editors of journals and books are mostly concerned that authors use terms that are understandable within the context of single publications (disciplines). Even if review journals (e.g., Trends in Ecology and Evolution) encourage glossaries of terms, authors are given freedom to make up their own definitions – change but one word and the entire meaning of a concept can change considerably.
Now, most of the DD terms we revise in our review paper can indeed be understood within individual publications, but is that enough? We don’t think so. Regulated terminology (excuse the pun) is taken as a logical necessity in fields such as astronomy, biochemistry, medicine and taxonomy, because unique names for a star, molecule, heart disease or species are critical for application, sharing and review of existing knowledge, and for avoiding redundant research. Terminology merits due recognition because it is the basis of the classification of knowledge, and so indicates the state of progress and maturation of any science. A person can be the smartest scientist around, but without a clear command of language to communicate his/her ideas, that intelligence is wasted.
Ecological journals could improve terminological standards through permanent sections on terminological review, as well as guidelines whereby authors were obliged to authorise foundational definition of terms in their papers. Yet, neither terminological reviews nor editorial guidelines are panacea to solve the Ecological Tower of Babel, because reviews are subject to readership rates (so they can go unnoticed to many), and because researchers are in no way bound to terminological rules proposed by their peers. Terminology represents one of those matters about which many voices complain but see no solution.
The establishment of regulatory rules for ecological terminology once attempted by the Ecological Society of America24 is condemned to be an unpopular idea for many scientists. In a publication in preparation, we will argue that the creation of a ‘Code of Ecological Nomenclature’ – one that is endorsed by journals as a pre-requisite for manuscript acceptance – is the key for improving terminological standards and enforcing efficient communication in ecology. For the time being, we outline in Boxes 1 and 2 a few suggestions to prevent terminological confusion when writing a scientific manuscript, in particular, on density feedback.
| BOX 1 – Terminological suggestions for writing papers on density feedbacks– |
| Density feedback is preferable over ‘density dependence’, because ‘feedback’ encapsulates causality (inherent to the causal effects of changing population size on demographic rates), while ‘dependence’ indicates correlation25 |
| The qualifiers of density feedback compensatory/depensatory are preferable because they denote effects on population change free of statistical connotation. Avoid historical terms (i.e., ‘direct’, ‘inverse’, ‘negative’, ‘positive’) that confuse statistical signs (positive, negative) with density effects on population change (compensatory or depensatory) |
| Allee effect is a kind of depensatory density feedback operating only at low numbers (please let us know who coined the term ‘Allee effect’!). Delayed compensatory feedback represents a density feedback over more than two generations, years or sampling times. Overcompensatory feedback represents a strong density feedback that causes chaotic dynamics – but, to state that a density feedback is overcompensatory, we also need a metric of population variance. Be aware that fishery ecologists and botanists use ‘overcocompensation’ in other ways |
| ‘Density dependence’ and ‘regulation’ ARE NOT synonyms. To skip the sterile, historical debate about population regulation, avoid the term ‘density-dependent regulation’ (and variants), or even better: do not use the term ‘regulation’ at all. Moderate compensatory density feedback is only one requirement for population regulation. Density dependence/feedback does have a clear definition and well-established methods of measurement. But what does regulation means? How can it be measured? Why is it important? |
| The expressions ‘ensemble’ and ‘component’ density feedbacks distinguish feedbacks on a population’s overall growth rate (ensemble) or on one of its rates of fertility, survival or dispersal (component). To appreciate the importance of such distinction, bear in mind the theoretical principle that an ensemble density feedback is the result of the compound interplay of all component density feedbacks. Thus, detectable component feedbacks might contribute nothing to ensemble density feedbacks, and are certainly no indication of population regulation26 |
| The terminology of density feedback distinguishing spatial and temporal population change needs urgent revision, particularly with regard to how zoologists and botanists have been investigating density feedbacks in both contexts. This conceptual friction is at the core of the old controversy between Andrewartha and Nicholson. This controversy is unresolved, despite the claims that it is no longer a problem. If we ask 100 ecologists to define ‘regulation’ and ‘limitation’ and yield > 10 substantially different versions (you would probably get more than that), something must be wrong! |
| BOX 2 – Terminological suggestions for writing scientific papers– |
| Explore who/when/where coined the key words of your manuscript |
| Provide definitions of the core concept you are examining (often named in the title of your study) and, if necessary, of the string of key-words |
| All definitions should be authorised, unless you are proposing a new concept or term, which should be clearly stated as such |
| Explain in the Methods how you have handled terminology where conflict persists. Of course, be consistent with your terminology throughout the main text and appendices, e.g., devote one read-through only to check your vocabulary |
| If, by the end of your manuscript, you think that the terminology of your area of research is inconsistent, write a review paper (be confident that it might even be highly cited!) |
| As a supervisor, encourage your students to include (and publish) a thesis chapter on terminology – Master/PhD students have the time to wade through the old and modern literature, while a historical revision of terminology is a secure way of getting an insight into the topic of an investigation across disciplines |
| As an editor, by all means be sure that a terminological manuscript is refereed fairly, and remember that our discipline needs this stuff urgently. Top review journals such as Trends in Ecology and Evolution or Annual Review – Ecology, Evolution, and Systematics have paid scanty attention to this problem, and could commission experts to revise terminology – bringing together experts who disagree seems like an interesting approach, and those who love their discipline (more than their egos) might be happy to tackle the challenge |
—
Salvador Herrando-Pérez & CJA Bradshaw
—
References
- A. R. E. Sinclair, in Ecological concepts: the contribution of ecology to an understanding of the natural world, edited by J. M. Cherret (Blackwell Scientific, Oxford, UK, 1989), pp. 197
- P. A. Stephens, W. J. Sutherland, Trends in Ecology and Evolution 14 (10), 401 (1999)
- S. J. Kutz, J. Ducrocq, G. G. Verocai et al., Advances in Parasitology 79, 99 (2012)
- M. S. Boyce, A. R. E. Sinclair, G. C. White, Oikos 87 (3), 419 (1999)
- K. A. Rose, J. H. Cowan, K. O. Winemiller et al., Fish and Fisheries 2, 293 (2001)
- F. Courchamp, T. Clutton-Brock, B. Grenfell, Trends in Ecology and Evolution 14 (10), 405 (1999)
- K. Henle, S. Sarre, K. Wiegand, Biodiversity and Conservation 13, 9 (2004); J. L. Sabo, E. E. Holmes, P. Kareiva, Ecology 85 (2), 328 (2004)
- W. F. Fagan, M. A. Lewis, M. G. Neubert et al., Ecology Letters 5 (1), 148 (2002); P. C. Tobin, L. Berec, A. M. Liebhold, Ecology Letters 14 (6), 615 (2011)
- A. A. Berryman, M. Lima, B. A. Hawkins, Oikos 100 (3), 636 (2003)
- G. C. Varley, Nature 181 (4626), 1780 (1958)
- S. H. Smith, Journal of Economic Entomology 28, 873 (1935)
- K. Shinozaki, T. Kira, Journal of the Institute of Polytechnics, Osaka City, Series D 7, 3 (1956)
- W. C. Allee, The American Naturalist 75 (760), 473 (1941)
- F. Neave, Journal of the Fisheries Research Board of Canada 9, 450 (1953)
- G. E. Belovsky, D. B. Botkin, T. A. Crowl et al., BioScience 54 (4), 345 (2004)
- J. B. S. Haldane, in New Biology, edited by M. L. Johnson, M. Abercrombie, G. E. Fogg (Penguin Books, London, 1953), Vol. 15, pp. 9
- W. W. Murdoch, S. J. Walde, in Toward a More Exact Ecology, edited by P. J. Grubb, J. B. Whittaker (Blackwell, Oxford, 1989), pp. 113
- L. Berec, E. Angulo, F. Courchamp, Trends in Ecology and Evolution 22 (4), 185 (2007)
- S. Herrando-Pérez, S. Delean, B. W. Brook, C. J. A. Bradshaw, Oecologia doi:10.1007/s00442-012-2347-3 (2012)
- B. M. Starzomski, B. J. Cardinale, J. A. Dunne et al., Ecological Society 9 (1), 14 (2004)
- M. Clavero, Trends in Ecology and Evolution 25 (10), 552 (2010); M. Clavero, Frontiers in Ecology and the Environment 9 (2), 93 (2011)
- J. C. Dauvin, G. Bellan, D. Bellan-Santini, Aquatic Conservation-Marine and Freshwater Ecosystems 18 (4), 446 (2008)
- D. D. Murphy, B. D. Noon, Journal of Wildlife Management 55 (4), 773 (1991); J. R. Weber, C. S. Word, BioScience 51 (6), 487 (2001); D. C. Adams, M. S. DiBitetti, C. H. Janson et al., Oikos 80 (3), 632 (1997)
- F. E. Eggleton, G. L. Clarke, H. A. Gleanon et al., Report of the committee on nomenclature of the Ecological Society of North America at the Cornell University meeting, September 1952 (1952); H. C. Hanson, J. G. Needham, W. P. Taylor et al., Science 74 (1930), 648 (1931)
- A. A. Berryman, M. Lima, B. A. Hawkins, Oikos 99 (3), 600 (2002)
- S. Herrando-Pérez, S. Delean, B. W. Brook, C. J. A. Bradshaw, Ecology 93 (7), 1728 (2012)
- M. Ferrer, V. Penteriani, Journal of Applied Ecology 45, 1453 (2008)
- B. B. Lamont, P. G. L. Klinkhamer, E. T. F. Witkowski, Oecologia 94 (3), 446 (1993)
- S. J. Holbrook, R. J. Schmitt, Ecology 83 (10), 2855 (2002)



[…] with the larger terminological quagmires of specific disciplines (ecology included), a clear, quantifiable definition should be the norm for any such terminology, especially if it is […]
LikeLike
[…] Ecology is a Tower of Babel (17/09/12,OR) / Terminology of density dependence […]
LikeLike
[…] debates of the 1970s and 1980s, and we’ve pilfered precious years arguing about whether density feedback actually exists (answer: it […]
LikeLike
[…] read, yet despite citing them for decades, few of us have (which has all sorts of implications for bullshit perpetuating over time, but that’s a topic for another blog post). For now, this is my […]
LikeLike
[…] I then experienced to understand the literature were caused by a formidable jargon comprising > 50 terms for naming only 4 DD types, and many associated concepts in population dynamics (like boundedness, determination, limitation, […]
LikeLike
Interesting, reminded me of a conceptually similar, but a bit older paper:
V. Grimm and Christian Wissel (1997) Babel, or the ecological stability discussions: an inventory and analysis of terminology and a guide for avoiding confusion. Oecologia, 109, 323-334.
Seems little has changed when it comes to the lack of clear definition for ecological terms.
LikeLike
[…] Fed up with linguistic tricks? Well, too bad, because here’s another one. It turns out you can use agricultural biodiversity terminology as examples to explain what’s wrong with ecology. […]
LikeLike
I actually tried to find who coined the term ‘Allee effect’ several weeks ago, and the earliest use of the term I found was Gordon RE (1952) A contribution to the life history and ecology of the Plethodontid Salamander Aneides aeneus. American Midland Naturalist 47(3), 666-701.
Not sure if that helps, but thanks for the article, Salva and Corey.
LikeLike